Our webinar on PBPK Modeling features speakers Dr. Steven Webb and Dr. David Cowie from Syngenta. Together, they will share models to assess how Tyrosine aminotransferase (TAT) catalyzes the conversion of tyrosine to accumulation of hydroxyphenylpyruvic acid (HPPA) and how its activity varies among mammalian species. Species differences also exist in the ability to utilize alternative pathways for tyrosine catabolism …
Integrating computational tools and human cell-based NAMs for human-relevant risk assessments
Our first September 2024 webinar focuses on novel methods to assess relevant risk in humans with Les Recio, PhD, DABT, ATS, and Rasim Barutcu, PhD. We believe these approaches are a positive and proven way forward for expediting research and reducing the use of animals. While we do not offer a training course for integrating these tools, we do offer …
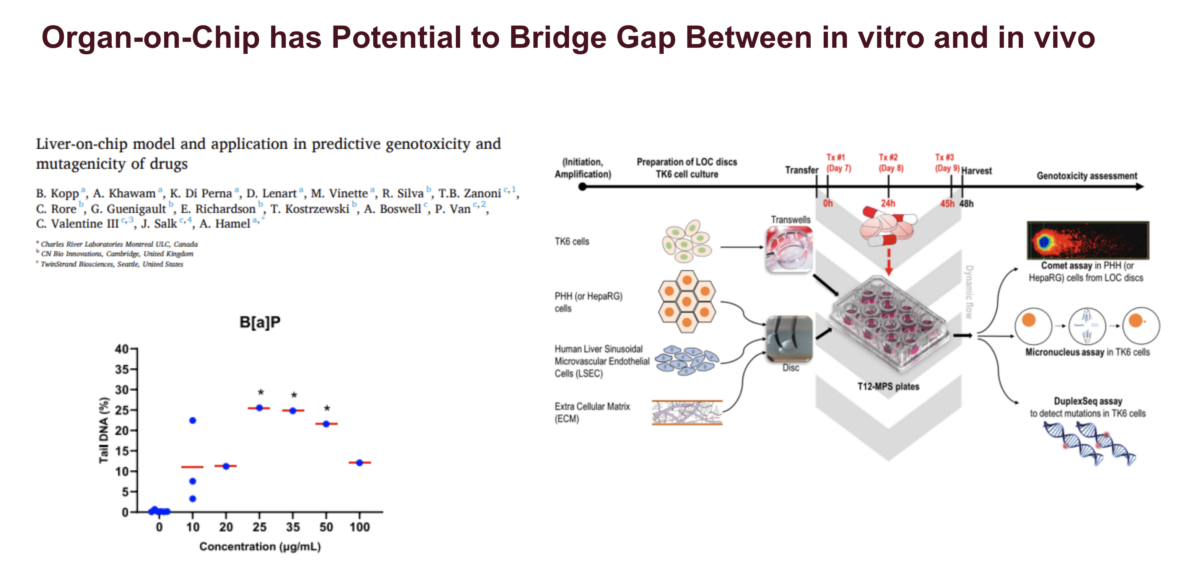
Quantitative Interpretation of in vitro Genotoxicity Data for Risk Assessment
If you attended /watched the recordings from the last two webinars, you might remember that Dr. Paul White and Dr. George Johnson highlighted the need to develop a conceptual framework to use genotoxicity data quantitatively for risk assessment. In this webinar, Marc Beal, PhD, a Research Scientist and Head of the Molecular and Applied Toxicology Section within the Food and …
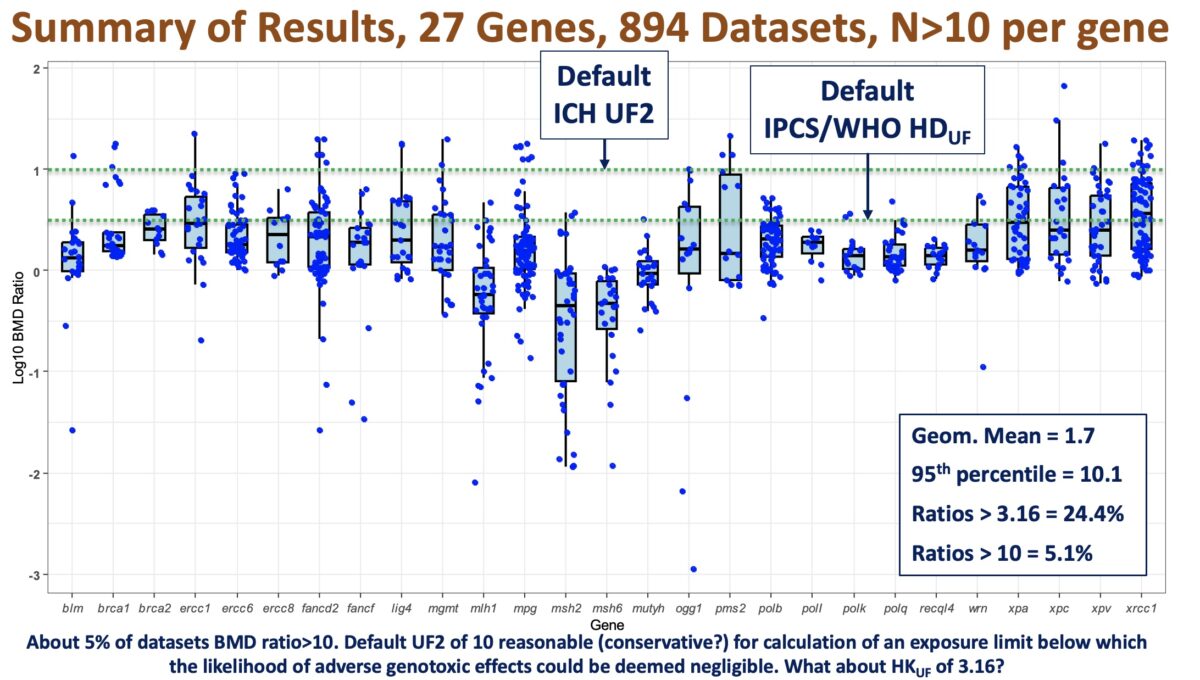
Quantitative Interpretation of Genetic Toxicity Dose-response Data for Risk Assessment and Regulatory Decision-making: Opportunities, Challenges and Future Prospects
In continuing the momentum on the topic of quantitative interpretation of genotoxicity dose-response data for risk assessment, we have the pleasure of hosting Paul A. White, PhD as our next webinar speaker. Dr White is a research scientist at the Environmental Health Science and Research Bureau of Health Canada in Ottawa, Ontario, Canada. What you will learn: Assumptions underlying the …
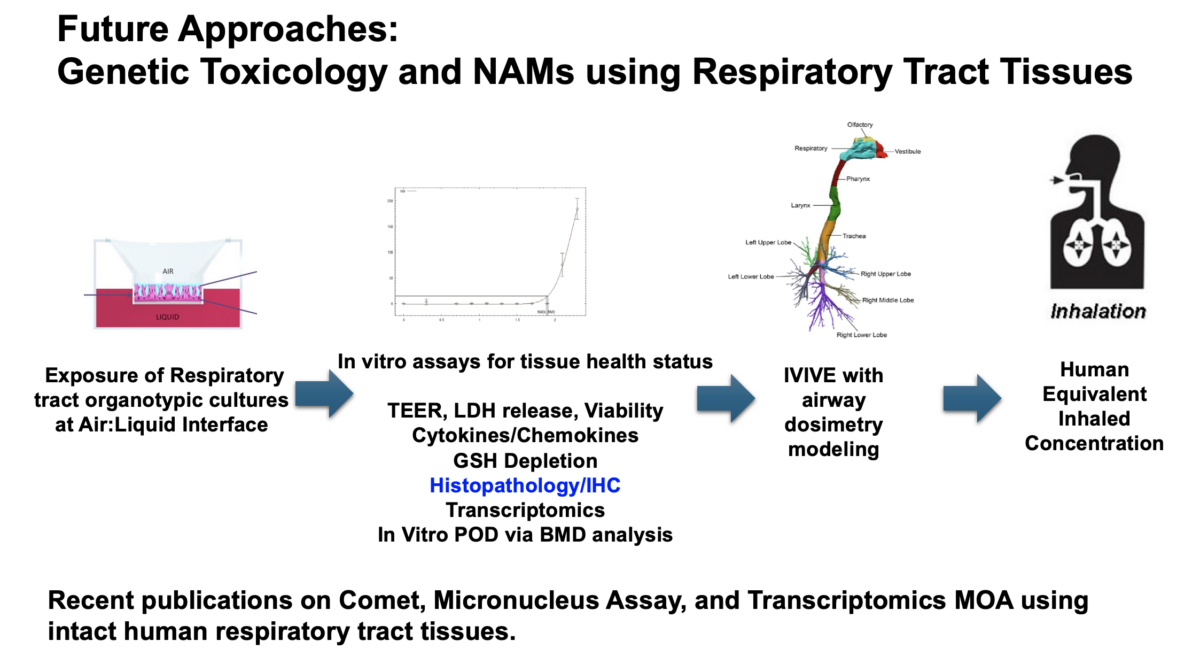
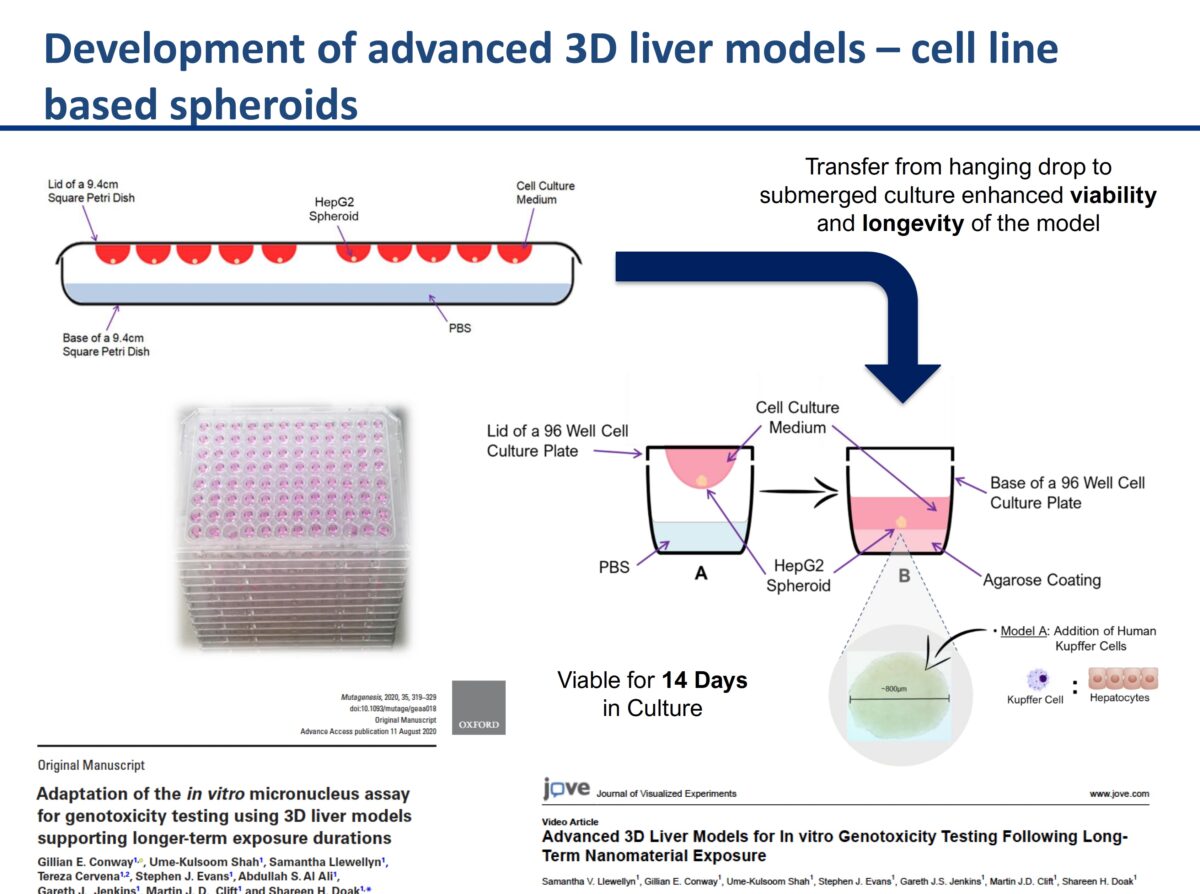
3D cell culture models & their role in enhancing genotoxicity testing approaches for nano- and advanced-materials
It was an honor to have Professor Shareen Doak with us to discuss her groundbreaking research. Her research on New Approach Methodologies (NAMs) and overcoming limitations of OECD Test Guidelines for nanomaterials (NMs) testing is leading edge brought up a lot of great questions. The link to the recording is below. What you’ll learn: The need to adapt existing …
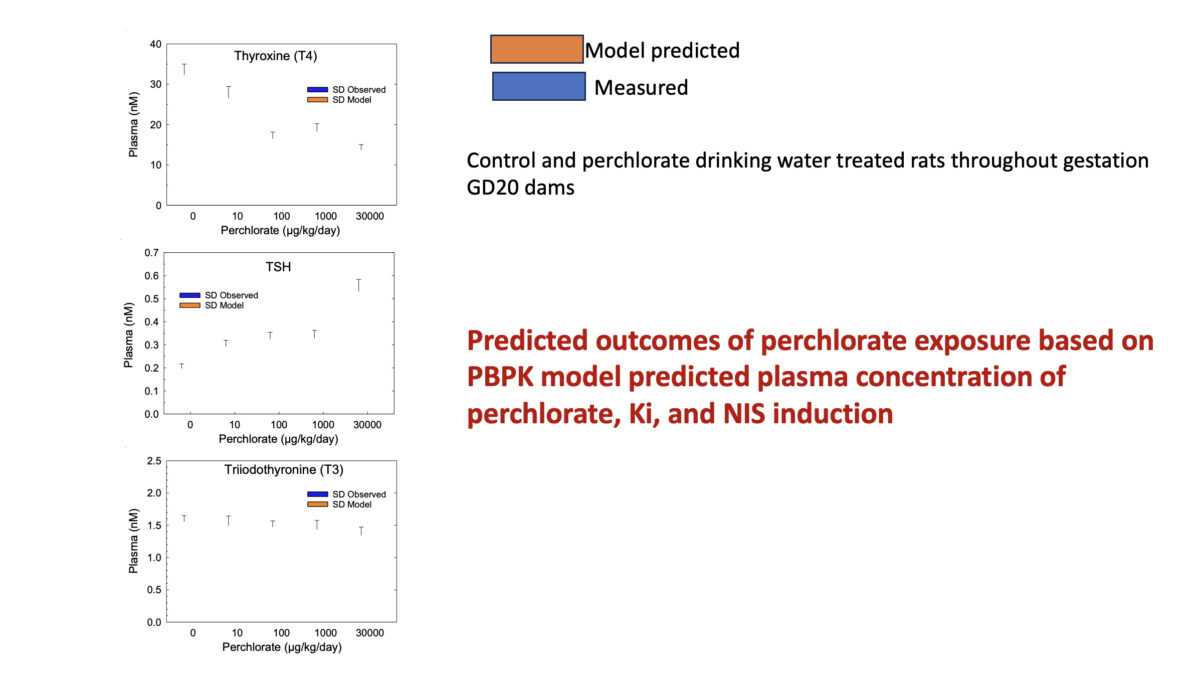
Towards Translating In Vitro Measures Of Thyroid Hormone System Disruption To In Vivo Responses In The Pregnant Rat Via A Biologically Based Dose Response (BBDR) Model
We are excited to present our first recorded installment for our 2024 webinar series. Jeff Fisher, PhD, ScitoVation’s Senior Science Fellow, will be speaking about thyroid disruption and IVIVE based on his latest publication. ScitoVation scientists continue advancing the benefits of IVIVE modeling. What you’ll learn: Thyroid modeling demonstrating IVIVE for a molecular initiating event determined in vitro How to …
Generic Pharmacokinetic Models for Mother-to-Offspring Transfer of Chemicals
Our speaker is Dustin Kapraun, PhD, physical scientist with the Center for Public Health and Environmental Assessment (CPHEA) in the Office of Research and Development (ORD) at the U.S. Environmental Protection Agency (EPA). In developing human health risk assessments, it is important to consider pregnant women, developing fetuses, and nursing children because chemical exposures experienced by these groups can lead …
ToxAIcology – the future of toxicology is AI
Artificial Intelligence (AI) is impacting all types of work, including toxicology. Dr Hartung will discuss how to move toxicology to a more holistic and integrated paradigm using AI and how AI plays a role in setting the direction of “Toxicology for the 21st Century 2.0” in future decades. What you’ll learn: Sources of Big Data for toxicology and various AI …
Physiologically based pharmacokinetic modeling as a data analysis and study design tool – case studies in nonlinear pharmacokinetics
We just had a wonderful talk from Dr. Dan Hoer. Dr Hoer is a physical scientist with the US Environmental Protection Agency, Office of Pesticide Programs. His talk will focus on non-linear PBPK modeling, and the advantages offered by this approach. What you’ll learn: Important advantages of PBPK modeling over statistical methods that are often restricted to bimodal distinctions of …