Mitochondrial Fragmentation
Mitochondria are dynamic organelles capable of changing their organization and shape based on intracellular and extracellular signals. By balancing cycles of fusion and fission, mitochondria can regulate their morphology. Mitochondria respond to certain stresses, such as loss of mitochondrial membrane potential, by shifting the balance towards fission, resulting in fragmented mitochondria. Fragmentation is characterized by a large number of smaller mitochondria rather than a network of highly interconnected and elongated mitochondria. Fragmentation enables cells to eliminate non-functional mitochondrial fragments.
Mitochondrial fragmentation. Mitochondria are filamentous under untreated conditions (left), but the drug treatment (right) causes mitochondrial fragmentation within 10 minutes.
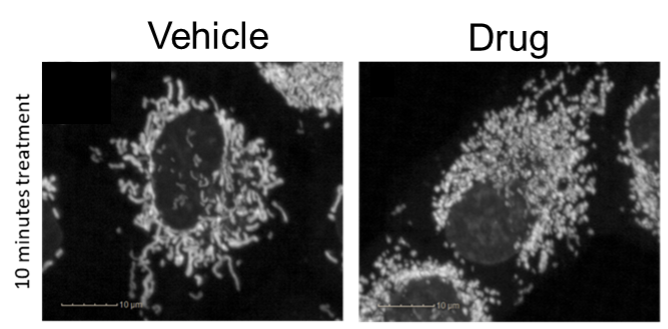
The mitochondrial fragmentation assay quantitates the fragmentation of mitochondria in response to compound treatment. In this assay, the mitochondria are stained with a dye, then treated with compound, and imaged live at high resolution. The images are analyzed on a per-cell basis to quantitate the balance between “spot” and “ridge” textures within the mitochondrial image. The output is a “fragmentation index”. An example of the use of the Mitochondrial Fragmentation Assay is shown below.
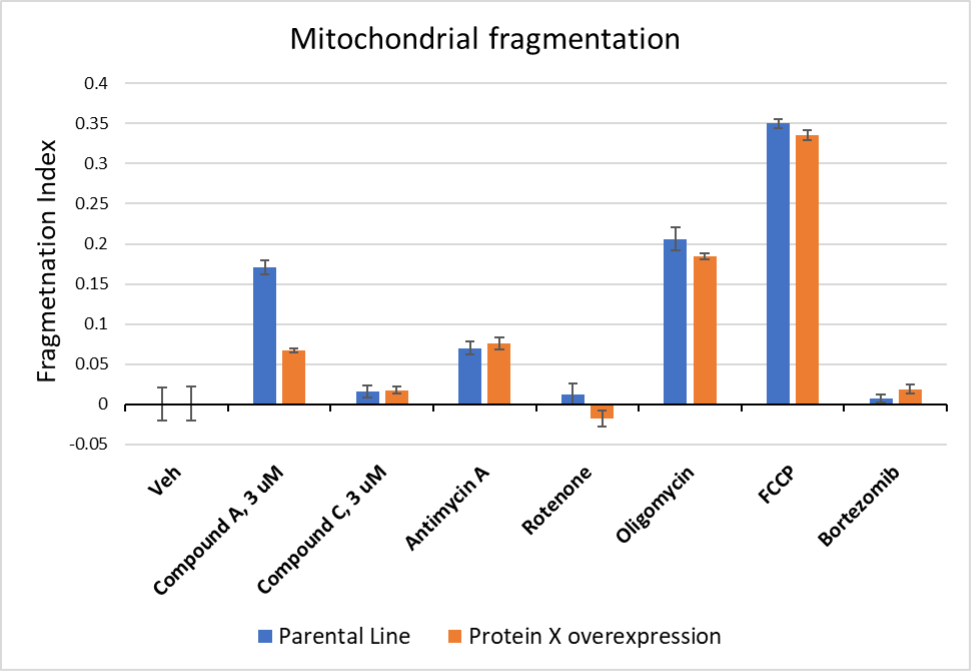
Mitochondrial Fragmentation Assay example. Two types of cells, a parental cancer cell line or a line derived from the parent by overexpression of Protein X, were exposed for 5 hours to test articles (Compounds A and C) or to control compounds with known effects. Control compounds known to disrupt mitochondrial function, including antimycin A, oligomycin, and FCCP, caused fragmentation relative to vehicle control with similar magnitudes in both cell lines. Bortezomib, which is a proteasome inhibitor, did not induce fragmentation. Compound A induced mitochondrial fragmentation in the parental cell line, but fragmentation was suppressed in cells with Protein X overexpression. Compound C did not induce fragmentation in either line.
